July 2022
Intermediate result from partners: CIDETEC, RWTH
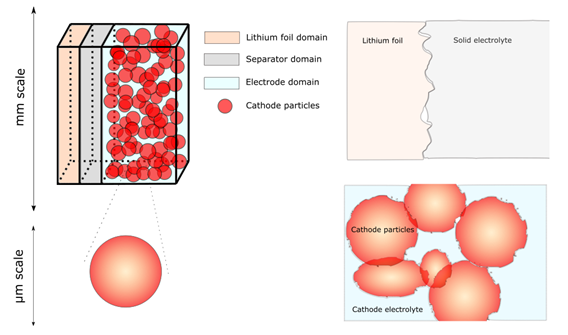
Interfaces play an important role in the dynamics of solid-state lithium-metal batteries. We have analyzed the main differences between the standard electrochemical models for liquid electrolytes and the response of solid electrolyte systems that add additional complexity to the modelling because of the variability of the properties depending the composition, and because of the more complex interplay at the solid-solid interfaces.
Read the public summary of the report here.
- Objective: The objective is to extend the standard electrochemical models used in literature and industry for liquid electrolyte systems, to solid-state electrolyte cells including lithium foil as anode.
- Research: We studied the variability of the electrolyte conductivity depending on the amount of polymer and ceramic filling to predict the optimum composition of the electrolyte. Additionally, we studied the mechanics at the different solid-solid interfaces, using homogenization theory and a phase field model to understand the details at the lithium interfaces.
- Result: The results were on one hand a simple model to adjust the electrolyte conductivity with experimental data, and on the other hand a full coupled electro-chemo-mechanical model able to simulate the deformation of each of the elements of the cell.
- What will it be used for: The results will be used to optimize the performance of the next generation composite electrolyte cells, by modifying their design. Additionally, some detailed models as the phase field model can be used to understand the underlying phenomenon and explore solutions for certain problems as formation and growth of lithium dendrites.
- Impact: The models provide understanding on the impact of certain components in the final battery performance; meaning that can directly impact on the reduction of the experimental load to meet and improve the battery cell design of future battery materials.